More Information
Submitted: February 03, 2026 | Accepted: February 12, 2026 | Published: February 13, 2026
Citation: Mukhiya G. The Role of CT and MRI in the Diagnosis of Orbital Tumors: A Literature Review. J Radiol Oncol. 2026; 10(1): 016-021. Available from:
https://dx.doi.org/10.29328/journal.jro.1001087
DOI: 10.29328/journal.jro.1001087
Copyright license: © 2026 Mukhiya G. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Keywords: CT and MRI; Diagnosis; Orbital tumors; Literature review
The Role of CT and MRI in the Diagnosis of Orbital Tumors: A Literature Review
Gauri Mukhiya*
Department of Radio-diagnosis and Interventional Radiology, B.P. Koirala Institute of Health Sciences, Dharan, Nepal
*Corresponding author: Dr Gauri Mukhiya, MBBS, MD, PhD., Department of Radio-diagnosis and Interventional Radiology, B.P. Koirala Institute of Health Sciences, Dharan, Nepal, Email: [email protected]
This literature review will help in the evaluation of orbital tumors using the CT and MR image features to determine if they have sufficient sensitivity to distinguish between benign and malignant tumors. These methods are important for diagnosing and assessing the location and extent of the pathological process before surgical exploration or other treatment interventions.
The orbit contains the globe, extra-ocular muscles, fat, vascular tissue, nerve, connective tissue and lacrimal gland. It is a small part of the skull, but various types of tumors can occur in the orbit, which may be primary, secondary or metastatic tumors. Orbital tumors are classified according to their site of origin and involvement of the adjacent tissues. Most Primary tumors arise inside the orbit with unknown causes of the lesions. Most of the primary tumors compress the optic nerve, and there is a deficit of vision or loss of vision, and it protrudes or pushes forward the globe, which is called proptosis. Proptosis is the most common sign of orbital tumors, masses and inflammatory conditions of the orbital structures.
Orbital masses are relatively uncommon compared to other masses in the body. It has been reported that two-thirds of orbital masses are benign and one-third of malignant tumors [1]. Characterization of orbital lesions plays an essential role in planning treatment strategies, as patient management varies greatly depending on the type of orbital lesion [2]. However, it is often challenging to differentiate between malignant masses due to their similar clinical presentations with proptosis. The accuracy of clinical diagnosis of orbital masses has greatly improved with CT and MRI imaging. MRI is a safe and noninvasive imaging method. It is best for soft tissues, the globe, nerves, blood vessels, and spatial resolutions. It can show the exact location, shape, margin, and size of lesions, and can also show secondary changes in the surrounding area of the lesions. CT is best for traumatic conditions of the orbit, bone and sinus-related diseases surrounding the orbit, bleeding, and calcification within lesions in the orbit. CT and MRI are widely used for orbital diseases. They are the modalities of choice for the assessment of orbital tumors. These conditions make a significant impact on characterization of orbital tumors and play an essential role in their management [1,3-5].
The CT and MR image features have sufficient sensitivity to distinguish between benign and malignant tumors. These methods are important for the diagnosis and assessment of the location and extent of the pathological process before surgical exploration or other medical treatments.
Anatomy of the orbit
The orbit contains a bony cavity, soft tissue structures, vascular structures, and nervous structures. The bony cavity is formed by: Roof: frontal bone and sphenoid bone. Medial wall: fontal process of the maxillary bone anteriorly, the lacrimal bone, the lamina papyracea of the ethmoid air cells, and the sphenoid bone posteriorly. Lateral wall: orbital surface of the zygomatic bone and sphenoid bone. Floor: orbital plate of the maxilla, orbital process of the palatine bone, and orbital surface of the zygomatic bone. Optic canal: It is located at the apex of the orbit and is bordered by two bony spurs of the lesser wing of the sphenoid bone. It contains the optic nerve and the ophthalmic artery.
The superior orbital fissure is located at the margin between the lateral wall and the orbital roof. It contains the superior and inferior ophthalmic veins, oculomotor nerve (iii), trochlear nerve (IV), abducens nerve (VI), and the ophthalmic division of the trigeminal nerve (V1). Inferior orbital fissure: It is located at the margin between the lateral wall and the orbital floor. Contents include: cranial nerve (V2), zygomatic nerve (branch of the pterygopalatine ganglion), and infero-orbital vessels (inferior ophthalmic vein).
The globe is the sensory organ of the orbit and represents the sense of sight of the eye. The globe is located within the orbit of the skull. It is protected by the bones of the orbit and allows movements of the eye muscles. It is innervated by the abducens nerve, oculomotor nerve and trochlear nerve. Pathological conditions of the globe of the eye include retinoblastoma, choroidal melanoma, glaucoma, and cataracts. It is essentially a spherical structure with a wall contains:-the retina (inner part), choroid (middle part), and sclera (outer part).
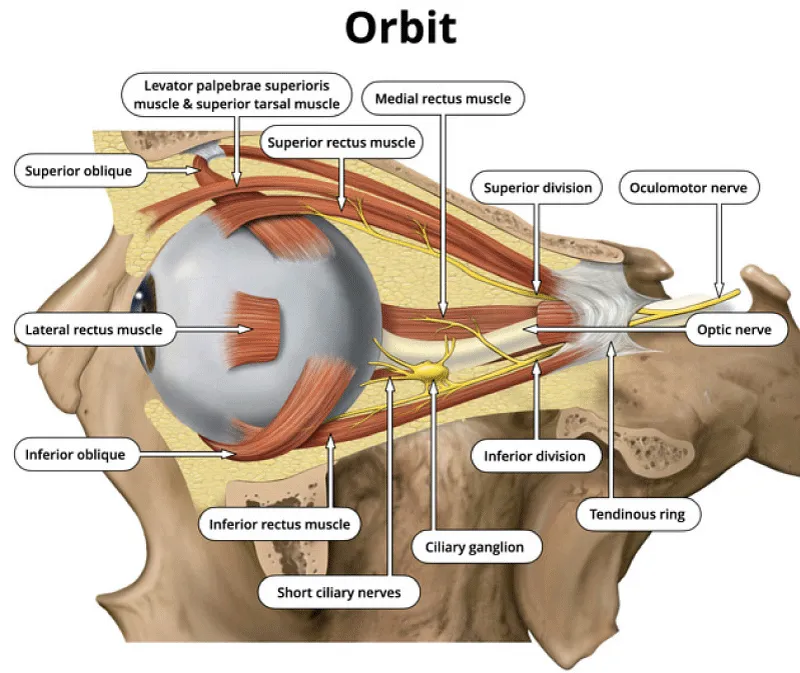
The globe is divided into three fluid-filled cavities: the anterior chambers, the posterior chamber, and the vitreous cavity [6,7]. The anterior and posterior chambers constitute the anterior segment, and the vitreous cavity constitutes the posterior segment of the orbit. The anterior chamber extends from the cornea to the iris, and the posterior chamber extends from the posterior surface of the iris to the anterior surface of the vitreous (Figure 1).
Figure 1: The Orbit.
There are seven extraocular muscles: superior rectus, superior oblique, inferior rectus, inferior oblique, medial rectus, levator palpebrae superior, and lateral rectus. The four rectus muscles and fibrous septa connect between them to form the muscle cone of the orbit. The muscle cone is divided into intraconal and extraconal spaces. The intraconal space is filled with the globe, optic nerve-sheath complex, orbital vessels, and nerves. The extraconal space contains the bony cage of the orbit, including the orbital wall, fat, and lacrimal gland. The intraconal space is located between the optic nerve and the extraocular muscles, while the extraconal space lies between the extraocular muscles and the orbital bony cage.
The primary arterial supply to the orbit is the ophthalmic artery, a branch of the internal carotid artery. It usually originates from the anteromedial aspect of the internal carotid artery as it exits the cavernous sinus. Secondary arterial supply to the orbit comes from the external carotid artery. The vascular anatomy in the orbit is well demonstrated on high-resolution magnetic imaging [8] and computed tomography angiography. The largest orbital vein visualized on CT or MRI is the superior ophthalmic vein. It arises near the base of the nose and drains into the cavernous sinus. The inferior ophthalmic vein is much smaller than the superior ophthalmic vein.
Imaging of the orbit
CT and MRI examinations provide complementary roles in orbital imaging. The choice between CT and MRI for initial imaging of the orbit depends on the clinical problem. CT is often preferred for trauma, bony orbit tissues, and calcifications, while MRI is generally preferred due to the absence of radiation risk and its high soft tissue contrast. MRI is the choice for evaluating the optic nerve, other cranial nerves and intracranial lesions. Sometimes, the exact diagnosis of meningiomas is missed by MRI, but CT can detect it better than MRI because meningiomas often present with calcifications.
MRI of the orbit can be performed with head coils for high spatial resolution imaging of the anterior orbital structures. For routine imaging, the field of view should include the cavernous sinus, optic chiasma, optic tract, nuclei of oculomotor, abducens nerve and trochlear nerve in the midbrain and pons. The protocol includes T1WI and T2WI in axial and coronal planes on MRI. Intravenous gadolinium contrast is routinely used. Fat suppression is usually performed for T2WI, post gadolinium imaging, to prevent the obscuration of enhancing lesions. Orbits are often included in routine CT screening examinations and usually performed according to the standard head or maxillofacial CT protocol. Coronal CT images are especially important in the cross-sectional evaluation of all intraorbital structures. Intravenous contrast CT is often used in the evaluation of inflammation, infection, neoplastic, and vascular orbital diseases.
There are many methods for the diagnosis of orbital tumors. The common principle is the localization of the orbital lesion, the four rectus muscles, the extraperiosteal space, and the optic nerve in the center of the four rectus muscles, the periosteal space, the lacrimal gland and the globe. The optic canal is located at the apex of the muscle cone, and the orbital septum is the baseline of orbital space [9,10].
MRI is superior to CT imaging for orbital diseases, but CT imaging is better than MRI in the assessment of trauma, bone lesions, sinus lesions, bleeding, and thyroid eye disease. Contrast enhancement is important for the diagnosis and differential diagnosis of orbital diseases. Some cases may require specific MRI scan sequences, such as fat suppression scans, FLAIR, GRE, DWI, CTA, and MRA.
CT and MR imaging play an essential role in identifying the features of orbital tumors and are important for managing orbital lesions [1,4,5]. CT and MRI studies are effective in assessing orbital lesions, with specific diagnostic criteria applied in the analysis of these tumors, including patient age, sex, size, shape, margin, density, CT value, signal intensity, contrast enhancement pattern and involvement of adjacent tissues and bone. CT and MRI images are crucial modalities for diagnosing and evaluating the location and extent of a pathological process before surgical exploration and for marking differential diagnoses [11,12].
Diagnosis and differential diagnosis of orbital tumors
There are various characteristics of an orbital lesion that can be used to help in differential diagnosis, including location, anatomic structure, imaging features, enhancement pattern, bony and adjacent tissue involvement around the lesion, age and chronicity of the lesion in patients [13]. The orbital tumor is one of the most common causes of blindness, so we should be aware of the examination and diagnosis of orbital lesions promptly to prevent blindness and chronic eye diseases. Orbital tumors are classified according to their site of origin and involvement of the adjacent tissues. The orbital lesions have a low incidence of diagnosis and treatment strategy due to several complications post- operative and treatment complications, such as cosmetic problems, severe ophthalmological deficits with loss of vision and eye movement issues [14]. The most common tumor in orbital lesions in adult patients is cavernous hemangioma, and the most common orbital lesion in children is rhabdomyosarcoma.
The muscle is composed of interconnected extraocular muscles to form a cone. The most common diseases are Graves’s disease and inflammatory pseudotumor. Graves’s disease is an autoimmune disease and the most common cause of bilateral exophthalmos. It is more commonly seen in females aged 40 to 50 than in males. Imaging findings most commonly involve hypertrophy of the inferior and medial rectus muscles (75%), followed by the superior rectus muscle (50%) and tendon involvement. CT and MRI sagittal section imaging show the length of the superior rectus and inferior rectus [15].
Another cause of hypertrophy is intraorbital inflammatory disease or orbital pseudotumor. Orbital inflammatory pseudotumor is a common tumor in the orbit, more commonly seen in individual aged10 to 40. It is a benign tumor, an idiopathic non-granulomatous inflammatory process that usually involves extraocular muscles and fat. It less frequently involves other intraorbital structures, including the uveal tract, sclera, optic nerve, and lacrimal gland [16,17]. It is usually a diffuse type of inflammatory orbital tumor. On CT, it shows slightly higher density and mild enhancement. MRI shows iso-intensity on T1WI and slightly high intensity on T2WI. Lymphoma shows unilateral or bilateral lesions and can be a primary or systemic disease.
It can occur in any space of orbit, including extraocular muscle hypertrophy, and can also be seen in lacrimal gland involvement [2]. Metastatic tumors can also be seen involving the extraconal muscles. Commonly, metastasis from breast cancer, lung cancer, prostate cancer and other parts of the body, such as bone and skin cancer or primary tumor pathology is conducive to making a diagnosis.
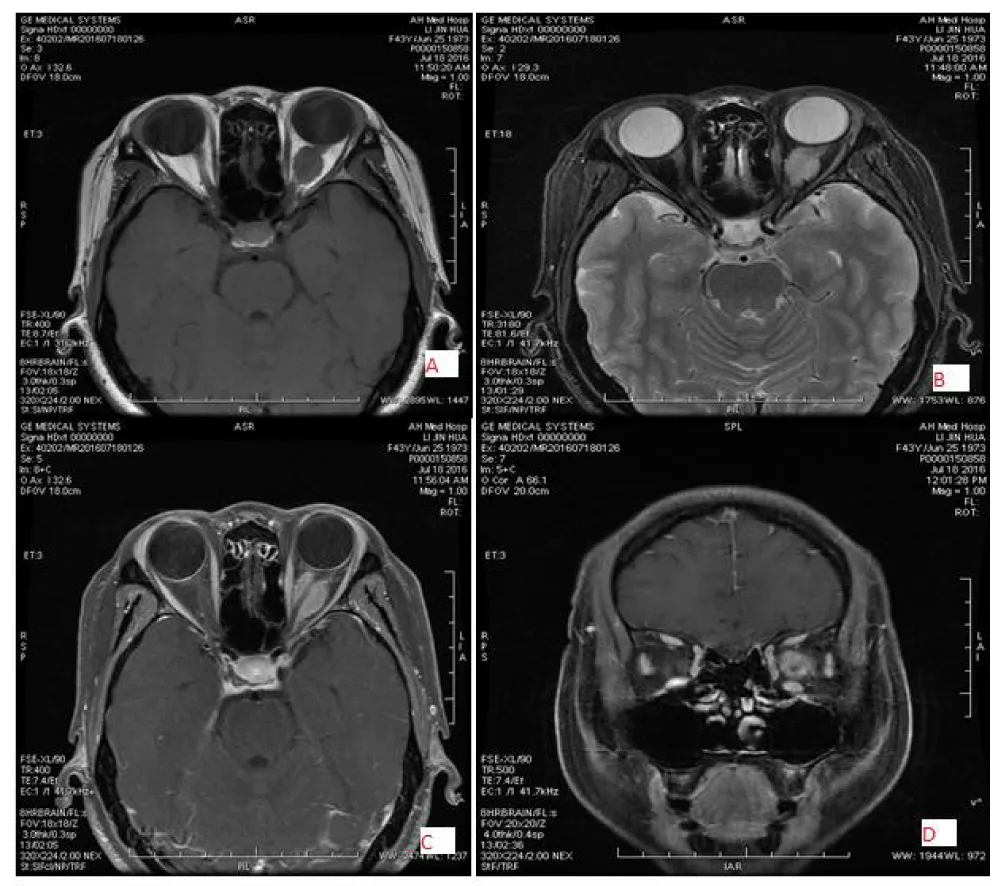
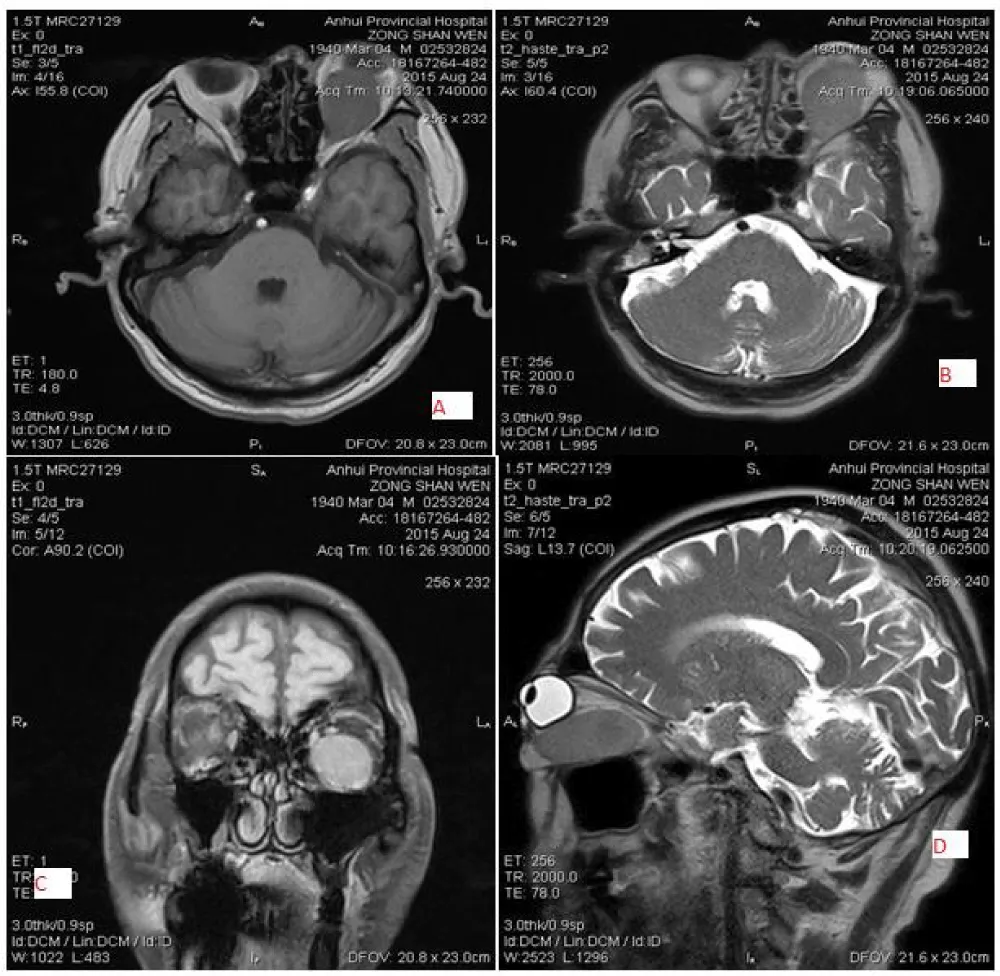
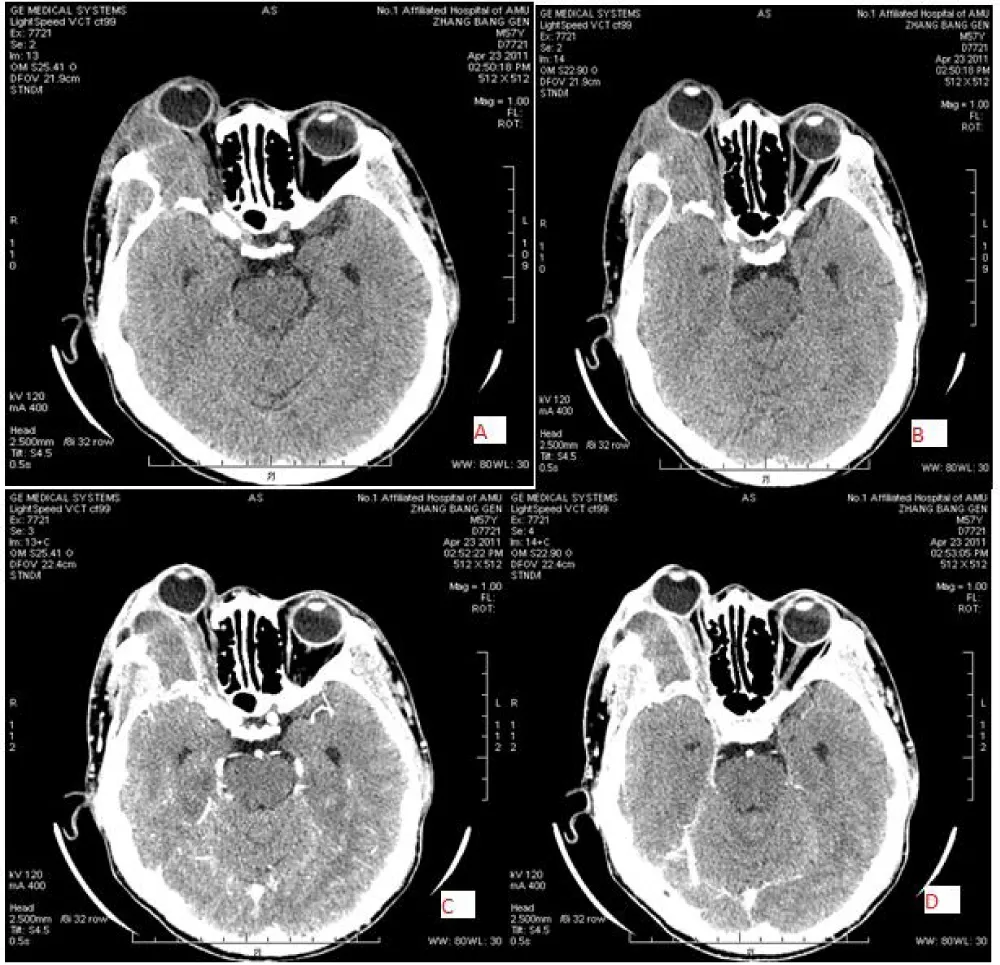
The most common primary optic nerve tumor is optic glioma in children under 5 years old (50%). Optic nerve glioma is a slow-growing, low-grade glioma tumor. 30% of patients with optic glioma have associated NF1, and those individuals have a better prognosis. Orbital meningioma is a slow-growing benign tumor that arises from arachnoid cap cells of the optic nerve sheath. Orbital meningioma can be distinguished from orbital lymphoma by showing less intense enhancement than meningioma, and by moulding around structures, which is present in lymphoma. Additionally, calcification is not seen in lymphoma, but it is frequently seen in orbital meningioma on CT images. Optic glioma is characterized by dilatation or enlargement of the optic nerve, while orbital schwannoma typically overlaps the optic nerve. In contrast, an orbital meningioma surrounds the optic nerve on CT and MRI scans (Figures 2,3).
Figure 2: 43-year-old female patient with left-sided meningioma, location surrounding the optic, well-defined and regular shaped, low signal intensity on T1WI (A) and slightly high signal intensity on T2WI(B) and marked homogeneous enhancement(C&D).
Figure 3: 44-year-old female patient with right-sided schwannoma, located in the intraconal and inferolateral region with well-defined margin and oval shape, low signal intensity on T1WI (A) and slightly high signal intensity on T2WI (B&C) and marked homogeneous enhancement (D).
Retinoblastoma is the most common primary malignancy originating from the retina in the eye globe. It often occurs in children under 5 years of age, can affect adults, and has the potential to metastasize. Retinoblastoma can be hereditary or non-genetic. Half of the patients with retinoblastoma present with a white pupil. A CT scan shows spotted calcifications, and routine magnetic resonance imaging screening is recommended during the first five years of life. Choroidal melanoma arises from the blood vessel layer of the choroid in the retina in the globe. It is a common primary malignant intraocular tumor and the second most common type of primary melanoma in the body. Choroidal melanoma is usually asymptomatic, presenting with painless, progressive visual loss and paracentral scotoma during routine orbit examination. It is typically seen as a dome or mushroom-shaped, well-circumscribed mass under the retinal pigment epithelium. Other tumors that can occur in the globe include choroidal hemangioma, retinal detachment, optic nerve drusen, choroidal osteoma, cataract, and astrocytic hamartoma.
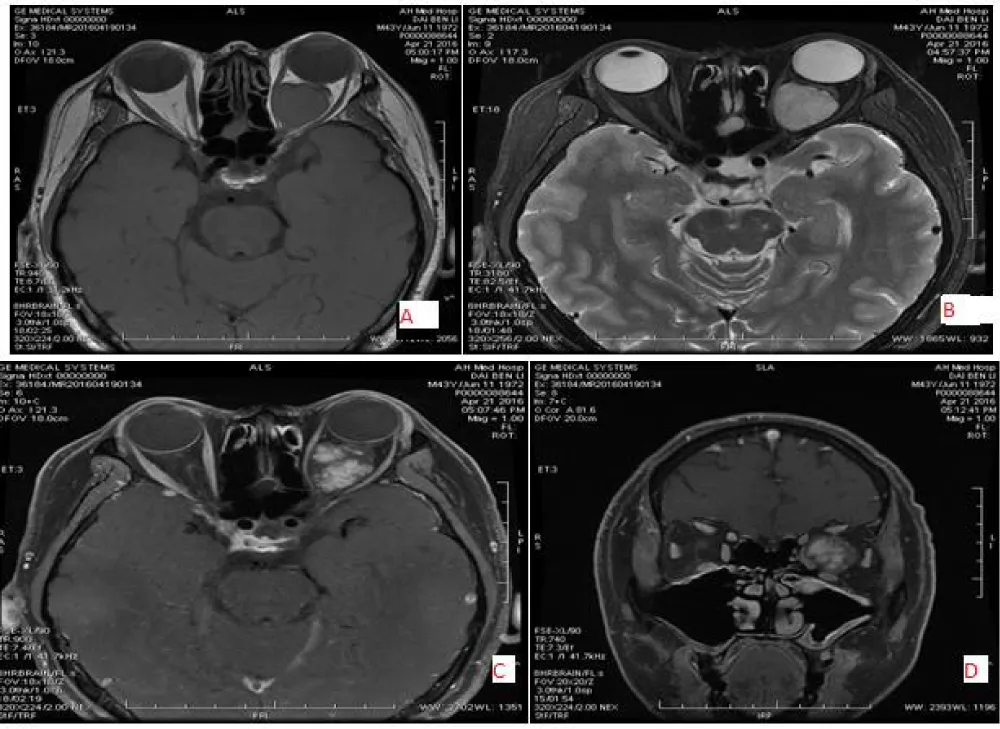
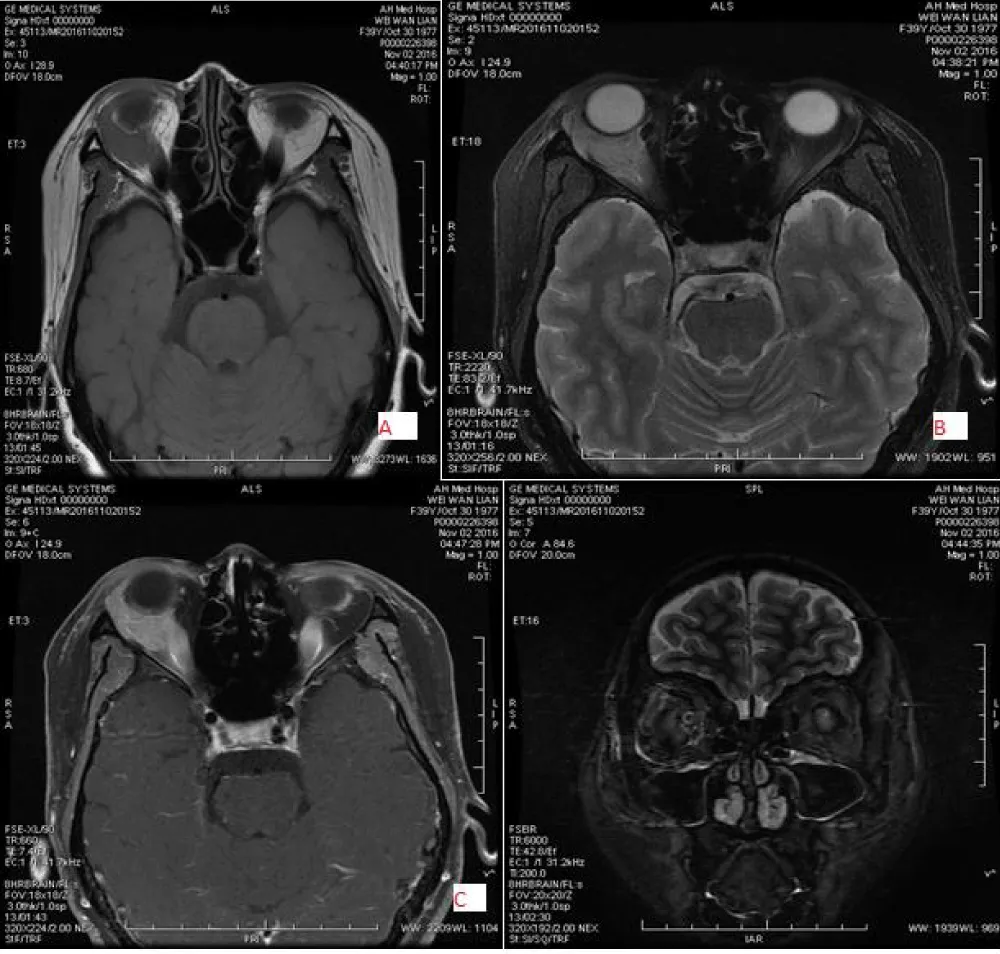
The intraconal space is located between the outside of the optic nerve and the inside of the extraocular muscle. In this space, many types of tumors may occur, but the most common tumor is cavernous hemangioma, seen in adults and capillary hemangioma in children. Cavernous hemangioma is usually present with a painless progressive mass, well-defined margin, regular shape, slightly high density, low or slightly high intensity on T1WI, high intensity on T2WI and gradual enhancement, making it easy to distinguish between the optic nerve and extraocular muscles. Orbital cavernous hemangioma and schwannoma present with slow progressive enlargement, and most of their clinical and radiological features are similar to each other [18-20]. Dynamic contrast-enhanced MRI is useful to distinguish between cavernous hemangioma and schwannoma. After Gadolinium administration in both patients cavernous hemangioma shows enhancement starting from a small point or portion and contrast media filling up the tumor, called “progressive enhancement”, while in the schwannoma, enhancement starts from a wide area and later shows heterogeneous enhancement. Other tumors that may occur in this space include schwannoma, meningioma, hemangiopericytoma, lymphoma, and metastasis (Figure 4).
Figure 4: 44-year-old male with left-sided cavernous hemangioma, located in the intraconal and superomedial region with oval shape and well defined, low signal intensity on T1WI(A) and high signal intensity on T2WI (B) and gradual homogeneous enhancement (C&D).
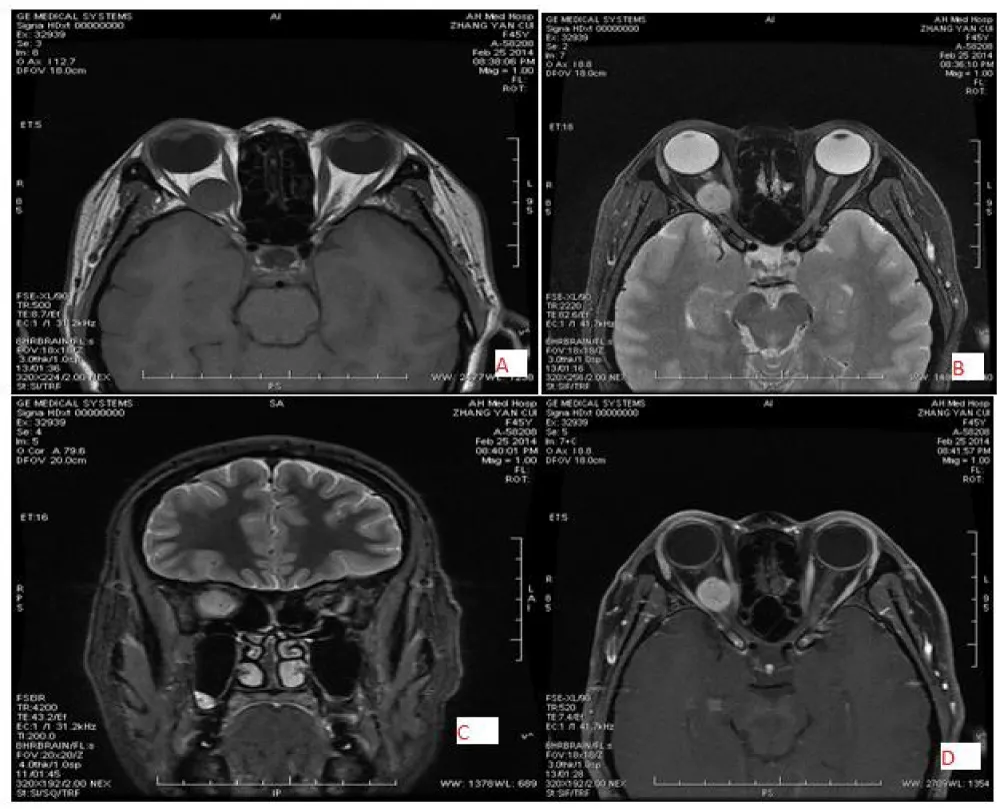
The extraconal space is located between the outside of the extraocular muscles and the inside of the orbital periosteum. In this space, mostly inflammatory and infectious diseases are seen. The inflammatory tumors include Graves’ disease, pseudotumor, granulomatous inflammation, and infectious diseases such as cellulitis and sinusitis. Orbital inflammatory pseudotumor is an idiopathic, benign, non-infectious inflammatory tumor in the orbit, with unknown causative factors. It presents with common signs and symptoms of a painful and unilateral mass, helping to distinguish it from thyroid-associated ophthalmopathy. It is usually a diffuse type of inflammatory disease and can be challenging to differentiate from other inflammatory diseases and neoplasms. However, a quick response to a trial of steroid therapy may aid in the diagnosis. Orbital lymphoma can be challenging to distinguish from orbital pseudotumor. Lymphoma is typically seen as decreased density on delayed images in dual-phase spiral CT contrast, while orbital pseudotumor is mostly seen as increased density on delayed images in dual-phase CT contrast [21]. Orbital lymphoma typically shows a brighter DWI signal and lower apparent diffusion coefficient (ADC) than normal orbital structures, whereas orbital pseudotumor shows intermediate DWI and ADC signal similar to the normal lacrimal gland [22]. Other tumors that can be found in this area include pleomorphic adenoma, adenoid cystic carcinoma, non- Hodgkin’s lymphoma, metastasis, and abscess (Figures 5-7).
Figure 5: 75-year-old male with left-sided lymphoma, located in the extraconal and inferomedial region with a regular-shaped and well-margined, slightly low signal intensity on T2WI (A) and iso intensity on T2WI (B & D)and marked homogeneous enhancement (C).
Figure 6: 57-year-old male with right-sided lymphoma, located in the extraconal and superolateral region with an irregular mass and ill-defined with invaded bone, iso density (38 HU) [A&B} and slightly homogeneous enhancement(C&D).
Figure 7: 39-year-old female with left-sided Inflammatory pseudotumor, located in the extraconal and inferolateral region with irregular shape and well margin, low signal intensity on T1WI (A) and slightly high signal intensity on T2WI(B&D) and mixed with intermediate enhancement (C).
This literature review will help in the evaluation of orbital tumors using the CT and MR image features to determine if they have sufficient sensitivity to distinguish between benign and malignant tumors. These methods are important for diagnosing and assessing the location and extent of the pathological process before surgical exploration or other treatment interventions.
Submission declaration
I declare that this manuscript is an original work and has not been published before, and is not currently being considered for publication elsewhere.
- Lemke AJ, Kazi I, Felix R. Magnetic resonance imaging of orbital tumors. Eur Radiol. 2006;16(10):2207–19. Available from: https://doi.org/10.1007/s00330-006-0227-0
- Valvassori GE, Sabnis SS, Mafee RF, Brown MS, Putterman A. Imaging of orbital lymphoproliferative disorders. Radiol Clin North Am. 1999;37(1):135–50, x–xi. Available from: https://doi.org/10.1016/s0033-8389(05)70083-x
- Mafee MF, Putterman A, Valvassori GE, Campos M, Capek V. Orbital space-occupying lesions: Role of computed tomography and magnetic resonance imaging. An analysis of 145 cases. Radiol Clin North Am. 1987;25(3):529–59. Available from: https://pubmed.ncbi.nlm.nih.gov/3575688/
- Warner MA, Weber AL, Jakobiec FA. Benign and malignant tumors of the orbital cavity including the lacrimal gland. Neuroimaging Clin N Am. 1996;6(1):123–42. Available from: https://pubmed.ncbi.nlm.nih.gov/8919138/
- Aviv RI, Miszkiel K. Orbital imaging: Part 2. Intraorbital pathology. Clin Radiol. 2005;60(3):288–307. Available from: https://doi.org/10.1016/j.crad.2004.05.018
- Belden CJ. MR imaging of the globe and optic nerve. Magn Reson Imaging Clin N Am. 2002;10(4):663–78. Available from: https://doi.org/10.1016/s1064-9689(02)00018-1
- Mafee MF, Karimi A, Shah J, Rapoport M, Ansari SA. Anatomy and pathology of the eye: Role of MR imaging and CT. Neuroimaging Clin N Am. 2005;15(1):23–47. Available from: https://doi.org/10.1016/j.nic.2005.02.005
- Ettl A, Kramer J, Daxer A, Koornneef L. High resolution magnetic resonance imaging of neurovascular orbital anatomy. Ophthalmology. 1997;104(5):869–77. Available from: https://doi.org/10.1016/s0161-6420(97)30219-x
- Bloching M, Beck R, Knipping S, Mir-Salim PA, Duncker GI, Berghaus A. [Orbital space-occupying lesions. Practical aspects of imaging]. HNO. 2001;49(1):21–8. Available from: https://doi.org/10.1007/s001060050703
- Wichmann W, Muller-Forell W. Anatomy of the visual system. Eur J Radiol. 2004;49(1):8–30. Available from: https://doi.org/10.1016/j.ejrad.2003.11.001
- Weber AL, Sabates NR. Survey of CT and MR imaging of the orbit. Eur J Radiol. 1996;22(1):42–52. Available from: https://doi.org/10.1016/0720-048x(96)00737-1
- Berletti R, Cavagna E, Cimini N, Moretto G, Schiavon F. [Dissection of epiaortic vessels: Clinical appearance and potentiality of imaging techniques]. Radiol Med. 2004;107(1-2):35–46. Available from: https://pubmed.ncbi.nlm.nih.gov/15031695/
- Koeller KK, Smirniotopoulos JG. Orbital masses. Semin Ultrasound CT MR. 1998;19(3):272–91. Available from: https://doi.org/10.1016/s0887-2171(98)90012-9
- Ohtsuka K, Hashimoto M, Suzuki Y. A review of 244 orbital tumors in Japanese patients during a 21-year period: Origins and locations. Jpn J Ophthalmol. 2005;49(1):49–55. Available from: https://doi.org/10.1007/s10384-004-0147-y
- Kahaly GJ. Imaging in thyroid-associated orbitopathy. Eur J Endocrinol. 2001;145(2):107–18. Available from: https://doi.org/10.1530/eje.0.1450107
- Flanders AE, Mafee MF, Rao VM, Choi KH. CT characteristics of orbital pseudotumors and other orbital inflammatory processes. J Comput Assist Tomogr. 1989;13(1):40–7. Available from: https://doi.org/10.1097/00004728-198901000-00008
- McNicholas MM, Power WJ, Griffin JF. Idiopathic inflammatory pseudotumour of the orbit: CT features correlated with clinical outcome. Clin Radiol. 1991;44(1):3–7. Available from: https://doi.org/10.1016/s0009-9260(05)80216-8
- Ruchman MC, Flanagan J. Cavernous hemangiomas of the orbit. Ophthalmology. 1983;90(11):1328–36. Available from: https://doi.org/10.1016/s0161-6420(83)34385-2
- Bilaniuk LT. Orbital vascular lesions. Role of imaging. Radiol Clin North Am. 1999;37(1):169–83, xi. Available from: https://doi.org/10.1016/s0033-8389(05)70085-3
- Gunduz K, Shields CL, Günalp I, Erden E, Shields JA. Orbital schwannoma: Correlation of magnetic resonance imaging and pathologic findings. Graefes Arch Clin Exp Ophthalmol. 2003;241(7):593–7. Available from: https://doi.org/10.1007/s00417-003-0681-1
- Moon WJ, Na DG, Ryoo JW, Kim MJ, Kim YD, Lim DH. Orbital lymphoma and subacute or chronic inflammatory pseudotumor: Differentiation with two-phase helical computed tomography. J Comput Assist Tomogr. 2003;27(4):510–6. Available from: https://doi.org/10.1097/00004728-200307000-00010
- Cytryn AS, Putterman AM, Schneck GL, Beckman E, Valvassori GE. Predictability of magnetic resonance imaging in differentiation of orbital lymphoma from orbital inflammatory syndrome. Ophthal Plast Reconstr Surg. 1997;13(2):129–34. Available from: https://doi.org/10.1097/00002341-199706000-00007